Center for Communications, Health and the Environment

E-Cigs Come Under Scrutiny
Electronic cigarettes are the new unknown in the war on tobacco. In the global marketplace for less than a decade and America since about 2007, these hand-held battery-operated products mimic the act of smoking, delivering nicotine, flavor and other chemicals via a vapor inhaled by the user through devices that often resemble cigarettes, cigars and pipes.
While their nicotine is derived from tobacco, e-cigarettes are not tobacco products, they are not cessation devices, and they are not FDA-approved.* In fact, even though they contain no tobacco, in April 2011, the U.S. Food and Drug Administration (FDA) announced that it will regulate e-cigarettes as tobacco products under the 2009 Family Smoking Prevention and Tobacco Control Act, after the courts blocked the agency from regulating them as drug-delivery devices.
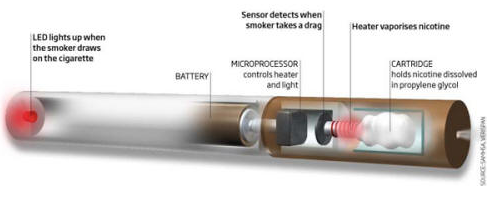 |
E-cigarettes may be easy to diagram, but experts debate their safety and use. (Source: e-cigarette-review.net) |
Harmless or Hazardous?
Various sources estimate e-cigarette use to be in the millions worldwide, with approximately 1 million “vapers” in the United States alone. To date, however, no specific FDA action has occurred, and consumers have little way of knowing whether e-cigarettes, currently sold through hundreds of small distributors online and in shopping mall kiosks, are safe for their intended use – and what that intended use is.
In their December 2010 article on the subject in the Journal of Public Health Policy, Zachary Cahn and Michael Siegel, M.D., M.P.H., detail 16 studies that have extensively cataloged the ingredients of e-cigarette liquid and vapor. Noting trace amounts of tobacco-specific nitrosamines in two studies and one occurrence of diethylene glycol, they find that “...few, if any, chemicals at levels detected in electronic cigarettes raise serious health concerns...[and] a preponderance of the available evidence shows them to be much safer than tobacco cigarettes and comparable in toxicity to conventional nicotine replacement products.” (This last finding being something the FDA has failed to mention, according to the authors.)
More recently, a July 2011 online ScienceDaily article reports, “testing of e-cig cartridges reveals poor quality control, variability in nicotine content among brands, and deviations between label claims and cartridge content,” not to mention unreliable nicotine delivery. In fact, manufacturers sell e-cigarette cartridges with up to 20 milligrams (mg) of nicotine (the average cigarette contains about 9 mg and the NICOTROL® Inhaler 10 mg), while refill kits enable consumers to load used cartridges with replacement solutions at much higher doses, and with non-nicotine products, such as marijuana hash oil. At the same time, no one has studied the safety in humans of inhaling propylene glycol, the main component of the vaporizing liquid in e-cigarettes, over an extended period of time.
Driving the ScienceDaily piece, Nathan K. Cobb, M.D., and David B. Abrams, Ph.D., take a closer look at e-cigs in their article “E-Cigarette or Drug-Delivery Device? Regulating Novel Nicotine Products,” published in the July 21, 2011 issue of The New England Journal of Medicine. Their findings reveal that, despite marketing claims, the devices tested did not deliver nicotine into the arterial blood as tobacco smoke does, and therefore do not perform like real cigarettes. And this, in conjunction with fluctuating nicotine content and unpredictable delivery, may make them ineffective quitting aids. Instead, the authors contend, e-cigarettes may simply become “ ‘bridge product[s]’ that smokers use in places where traditional tobacco smoking is prohibited, thus perpetuating their addiction and use of real cigarettes...[and] as 'starter' product[s] for young people considering smoking, especially since the cartridges can be purchased over the Internet with tempting flavoring...”
Addiction Aid or Cessation Device?
“Although the use of electronic cigarettes by nonsmokers is a theoretical concern, there is no existing evidence that youths or nonsmokers are using the product,” assert Cahn and Siegel, who propose that “...electronic cigarettes could entice smokers who were not otherwise inclined, to attempt to quit.”
In their article entitled, “Electronic cigarettes as a harm reduction strategy for tobacco control: A step forward or a repeat of past mistakes?,” they offer evidence of two published studies that “suggests that electronic cigarettes are capable of reducing cigarette craving, but that the effect is not due exclusively to nicotine” – and may actually have something to do with smoking-related stimuli, which may give e-cigarettes “an advantage over traditional nicotine delivery devices.” In fact, they argue, since e-cigarettes are not tobacco products, “switching to [them] is not an alternative to smoking cessation, but rather a form of smoking cessation akin to long-term use of NRT [nicotine replacement therapy].”
Underlining the murky nature of the debate, Cobb and Abrams point out that, shortly after the FDA’s announcement to regulate e-cigarettes, tobacco giants Philip Morris and British American Tobacco purchased nicotine-inhaler technologies that promise arterial/pulmonary delivery. This move, the authors argue, suggests the appearance of future consumer products that could “dramatically increase the risk of addiction and abuse,” – or, as critics Cahn and Siegel cynically remark, that “tobacco harm reduction will necessarily benefit the tobacco industry regardless of what else might be achieved.”
Ongoing Debate
The current challenge for concerned parties is to determine an effective means of regulating all existing and future refined-nicotine products, including e-cigarettes and other inhaler devices. According to Cobb and Abrams, this would include strict development of and adherence to safety measures, consistent specifications, quality control and functional dose limitations. With a nod to regulations surrounding the sale and marketing of such products to minors, Cahn and Siegel focus on available data, concluding that e-cigarettes in particular are “safer than tobacco cigarettes,” “show tremendous promise in the fight against tobacco-related morbidity and mortality” and “may fundamentally alter the tobacco harm reduction debate...[b]y dramatically expanding the potential for harm reduction strategies to achieve substantial health gains.”
Until research offers additional, actionable data or government agencies further control and clarify the production and use of such products, however, those looking for nicotine delivery devices may prefer already regulated options, such as patches, gum, lozenges, nasal spray, and the FDA’s own nicotine inhalation system.
* With the exception of the FDA-developed NICOTROL® Inhaler
Read More:
Lead Article: New WHO Report Projects Hope and Help in Global Tobacco Offensive
Spotlight Article: New Graphic Warnings Could Have Great Impact - IF Enacted
CECHE News: CECHE Champions Tobacco Control and Intervention Programs Around the World

Dr. Sushma Palmer, Program Director
Valeska Stupak, Editor & Design Consultant
Shiraz Mahyera, Systems Manager
Daniel Hollingsworth, Website Consultant